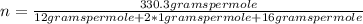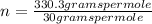Chemistry, 08.01.2020 22:31 jet0120996
The empirical formula for sucrose is ch2o. the molar mass of sucrose is 330.3 grams per mole. determine the molecular formula of sucrose.

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 22:00
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1


Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3
You know the right answer?
The empirical formula for sucrose is ch2o. the molar mass of sucrose is 330.3 grams per mole. determ...
Questions


Mathematics, 14.10.2019 12:20

Social Studies, 14.10.2019 12:20

Chemistry, 14.10.2019 12:20

Social Studies, 14.10.2019 12:20

Health, 14.10.2019 12:20

World Languages, 14.10.2019 12:20

History, 14.10.2019 12:20


Social Studies, 14.10.2019 12:20






Mathematics, 14.10.2019 12:20

Mathematics, 14.10.2019 12:20

Mathematics, 14.10.2019 12:20


History, 14.10.2019 12:20