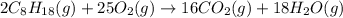Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3
You know the right answer?
2c8h18(g 25o2(g→16co2(g 18h2o(g how many moles of water are produced in this reaction...
Questions

Mathematics, 31.08.2019 15:50


Computers and Technology, 31.08.2019 15:50

Mathematics, 31.08.2019 15:50




Mathematics, 31.08.2019 15:50



English, 31.08.2019 15:50

Biology, 31.08.2019 15:50

Mathematics, 31.08.2019 15:50

Health, 31.08.2019 15:50


Health, 31.08.2019 15:50

Chemistry, 31.08.2019 15:50

Mathematics, 31.08.2019 15:50


Advanced Placement (AP), 31.08.2019 15:50