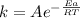Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2
You know the right answer?
If a temperature increase from 12.0 ∘c to 22.0 ∘c doubles the rate constant for a reaction, what is...
Questions


Mathematics, 31.10.2019 18:31

Social Studies, 31.10.2019 18:31


Physics, 31.10.2019 18:31

Biology, 31.10.2019 18:31

Spanish, 31.10.2019 18:31

Social Studies, 31.10.2019 18:31


Social Studies, 31.10.2019 18:31

Mathematics, 31.10.2019 18:31

World Languages, 31.10.2019 18:31

English, 31.10.2019 18:31

Mathematics, 31.10.2019 18:31

History, 31.10.2019 18:31


History, 31.10.2019 18:31



Biology, 31.10.2019 18:31