
Chemistry, 30.10.2019 17:31 msladycie8831
Which of the following is true in a basic solution?
a. the molarity of hydronium is double the molarity of hydroxide.
b. the molarity of hydronium is equal to the molarity of hydroxide.
c. the molarity of hydronium is lower than the molarity of hydroxide.
d. the molarity of hydronium is higher than the molarity of hydroxide.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3
You know the right answer?
Which of the following is true in a basic solution?
a. the molarity of hydronium is dou...
a. the molarity of hydronium is dou...
Questions

Chemistry, 19.10.2021 14:00


Mathematics, 19.10.2021 14:00

Social Studies, 19.10.2021 14:00

Social Studies, 19.10.2021 14:00

Chemistry, 19.10.2021 14:00

History, 19.10.2021 14:00

SAT, 19.10.2021 14:00


Advanced Placement (AP), 19.10.2021 14:00


Arts, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00


Engineering, 19.10.2021 14:00




Geography, 19.10.2021 14:00

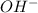 ions as compared to
ions as compared to 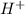 ions and an acidic solution has lesser number of
ions and an acidic solution has lesser number of 

