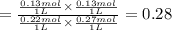Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1
You know the right answer?
In an experiment, 0.35 mol of co and 0.40 mol of h2o were placed in a 1.00-l reaction vessel. at equ...
Questions



Physics, 29.10.2020 01:00

Mathematics, 29.10.2020 01:00


History, 29.10.2020 01:00


Mathematics, 29.10.2020 01:00


Mathematics, 29.10.2020 01:00


Social Studies, 29.10.2020 01:00





English, 29.10.2020 01:00

History, 29.10.2020 01:00

Computers and Technology, 29.10.2020 01:00

Mathematics, 29.10.2020 01:00

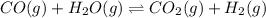
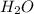 left at equilibrium = 0.40 mol - 0.13 mol = 0.27 mol
left at equilibrium = 0.40 mol - 0.13 mol = 0.27 mol![[CO]=\frac{0.22 mol}{1 L}](/tpl/images/0389/4768/0a0d1.png)
![[H_2O]=\frac{0.27 mol}{1 L}](/tpl/images/0389/4768/5bfa4.png)
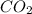 at equilibrium =
at equilibrium = ![[CO_2]=\frac{0.13 mol}{1 L}](/tpl/images/0389/4768/01e02.png)
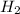 at equilibrium =
at equilibrium = ![[H_2]=\frac{0.13 mol}{1 L}](/tpl/images/0389/4768/fe16a.png)
![K=\frac{[CO_2][H_2]}{[CO][H_2]}](/tpl/images/0389/4768/437ec.png)