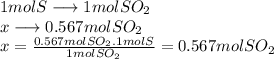The reaction: _s+o2=_so2
how many moles of sulfur must be burned to give 0.567 moles of so2?...

Chemistry, 19.09.2019 17:30 mohammedel04
The reaction: _s+o2=_so2
how many moles of sulfur must be burned to give 0.567 moles of so2?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1


Chemistry, 22.06.2019 14:40
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
You know the right answer?
Questions



Biology, 06.10.2019 19:00

Biology, 06.10.2019 19:00


Mathematics, 06.10.2019 19:00

Biology, 06.10.2019 19:00

Arts, 06.10.2019 19:00


Mathematics, 06.10.2019 19:00


Business, 06.10.2019 19:00


Mathematics, 06.10.2019 19:00


Social Studies, 06.10.2019 19:00

Business, 06.10.2019 19:00


History, 06.10.2019 19:00

Chemistry, 06.10.2019 19:00

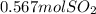

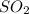 (we know it by the coefficients of balancing of the equation, which in this are equal to 1)
(we know it by the coefficients of balancing of the equation, which in this are equal to 1)