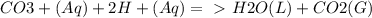Chemistry, 21.09.2019 08:50 yarrito20011307
Every antacid contains one or more ingredients capable of reacting with excess stomach acid (hcl). the essential neutralization products are co2 and/or h2o. write net ionic equations to represent the neutralizing action of the following popular antacids:
-rolaids
-maalox
-tums
-milk of magnesia

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
Every antacid contains one or more ingredients capable of reacting with excess stomach acid (hcl). t...
Questions

Mathematics, 08.01.2021 01:20





Biology, 08.01.2021 01:20

History, 08.01.2021 01:20

Biology, 08.01.2021 01:20


Mathematics, 08.01.2021 01:20

Mathematics, 08.01.2021 01:20




Mathematics, 08.01.2021 01:20



Physics, 08.01.2021 01:20


Mathematics, 08.01.2021 01:20