

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 01:00
Which elements are found in glucose, the product of photosynthesis? a. carbon, hydrogen, and oxygen b. carbon and hydrogen c. carbon, nitrogen, and oxygen d. hydrogen, nitrogen, and carbon
Answers: 2

Chemistry, 23.06.2019 04:00
What are the names of these two interactions with cattle and how do they differ from each other
Answers: 3

Chemistry, 23.06.2019 06:00
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes? or no? this question is worth 20 points! let it be correct!
Answers: 1
You know the right answer?
The fuel used in many disposable lighters is liquid butane, c4h10. butane has a molecular weight of...
Questions

Mathematics, 11.10.2019 04:00

Mathematics, 11.10.2019 04:00

Health, 11.10.2019 04:00

Mathematics, 11.10.2019 04:00


English, 11.10.2019 04:00

English, 11.10.2019 04:00

History, 11.10.2019 04:00

Mathematics, 11.10.2019 04:00







Physics, 11.10.2019 04:00

Mathematics, 11.10.2019 04:00

Mathematics, 11.10.2019 04:00


English, 11.10.2019 04:00

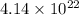
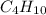 = 58.1 g/mole
= 58.1 g/mole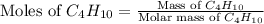
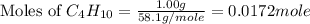
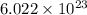 number of atoms.
number of atoms.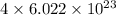 number of carbon atoms.
number of carbon atoms.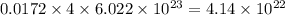 number of carbon atoms.
number of carbon atoms.

