
Determine the concentrations of k2so4, k , and so42– in a solution prepared by dissolving 1.80 × 10–4 g k2so4 in 1.50 l of water. express all three concentrations in molarity. additionally, express the concentrations of the ionic species in parts per million (ppm). note: determine the formal concentration of so42–. ignore any reactions with water.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1



Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1
You know the right answer?
Determine the concentrations of k2so4, k , and so42– in a solution prepared by dissolving 1.80 × 10–...
Questions



Mathematics, 27.09.2019 21:30

Mathematics, 27.09.2019 21:30

Physics, 27.09.2019 21:30

History, 27.09.2019 21:30

Social Studies, 27.09.2019 21:30

Health, 27.09.2019 21:30

Mathematics, 27.09.2019 21:30


Physics, 27.09.2019 21:30

Mathematics, 27.09.2019 21:30

Social Studies, 27.09.2019 21:30





Mathematics, 27.09.2019 21:30


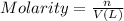
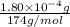
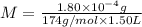
![[K_2SO_4]=M=6.8965\times 10^{-7} mol/L](/tpl/images/0431/3131/1bbe3.png)
![[K^+]=2\times M=2\times 6.8965\times 10^{-7} mol/L=1.3793\times 10^{-6} mol/L](/tpl/images/0431/3131/f96a0.png)
![[SO_4^{2-}]=1\times 6.8965\times 10^{-7} mol/L=6.8965\times 10^{-7} mol/L](/tpl/images/0431/3131/472ff.png)
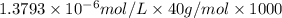
![[K^+]=0.05517 ppm](/tpl/images/0431/3131/6868b.png)
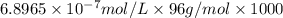
![[SO_4^{2-}]=0.06620 ppm](/tpl/images/0431/3131/956d0.png)


