Answer : The correct option is, (B) 0.72 mole of NaCl
Solution : Given,
Mass of NaCl = 42 g
Molar mass of NaCl = 58.44 g/mole
Formula used :
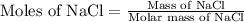
Now put all the given values in this formula, we get the moles of NaCl
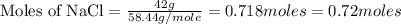
Therefore, the number of moles of NaCl is, 0.72 moles
























