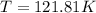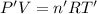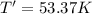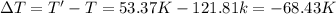Arigid, 2.50 l bottle contains 0.458 mol he. the pressure of the gas inside the bottle is 1.83 atm. if 0.713 mol ar is added to the bottle and the pressure increases to 2.05 atm, what is the change in temperature of the gas mixture? the initial temperature of the gas is k.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:50
If a substance is not at its melting or boiling point, as the heat content of a sample of matter increases, its temperature increases the number of intermolecular bonds decreases the space between particles increases the particles move faster
Answers: 2

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 17:00
The biosphere of the earth is made up of what compound? organic or inorganic?
Answers: 3
You know the right answer?
Arigid, 2.50 l bottle contains 0.458 mol he. the pressure of the gas inside the bottle is 1.83 atm....
Questions

English, 22.06.2019 20:30


Mathematics, 22.06.2019 20:30





History, 22.06.2019 20:30

Mathematics, 22.06.2019 20:30











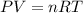 ( Ideal gas equation)
( Ideal gas equation)