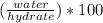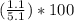Chemistry, 05.02.2020 13:01 andregijoe41
Asample of a hydrated compound has a mass of 5.1 g. during heating, it loses 1.1 g, leaving 4.0 g. what is the percentage by mass of water in the original hydrate?
6.11%
21.6%
27.5%
78.4%

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2

Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2

Chemistry, 22.06.2019 22:00
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1
You know the right answer?
Asample of a hydrated compound has a mass of 5.1 g. during heating, it loses 1.1 g, leaving 4.0 g. w...
Questions





Physics, 03.09.2020 15:01


Mathematics, 03.09.2020 15:01

Mathematics, 03.09.2020 15:01


Mathematics, 03.09.2020 15:01

Computers and Technology, 03.09.2020 15:01

Mathematics, 03.09.2020 15:01


History, 03.09.2020 15:01

Biology, 03.09.2020 15:01

Advanced Placement (AP), 03.09.2020 15:01

English, 03.09.2020 15:01