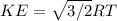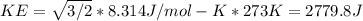Chemistry, 13.11.2019 01:31 raymond5799
What is the average kinetic energy of 1 mole of a gas that is at 0 degrees celsius?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3


Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
What is the average kinetic energy of 1 mole of a gas that is at 0 degrees celsius?...
Questions

History, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Mathematics, 13.10.2020 20:01

Physics, 13.10.2020 20:01




History, 13.10.2020 20:01



Mathematics, 13.10.2020 20:01






Biology, 13.10.2020 20:01