
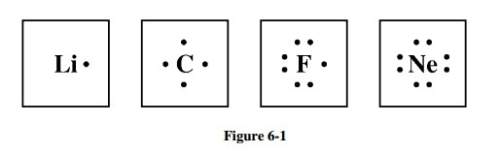
Study the electron dot diagrams for lithium, carbon, fluorine, and neon in figure 6-1. choose the statement that correctly identifies the most stable of the elements.
a. lithium is the most stable element because it has to lose only one electron to achieve a stable configuration.
b. carbon is the most stable element because it can form four bonds.
c. fluorine is the most stable element because it has to gain only one electron to achieve a stable configuration.
d. neon is the most stable element because its highest occupied energy level is filled.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:20
After watching the video "zinc strip in copper nitrate solution", and reading the instructions, click on the link labeled "start" just below the drawing of the pencil tip. follow the direction to complete the 3x3 grid. answer the below questions for the portion of the activity in which sn(s) is placed in agno3(aq)
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 23.06.2019 14:00
Which of the following is not a result when a change to an equilibrium system is applied? (2 points) increasing the rate of the forward reaction will cause a shift to the left. increasing the rate of the reverse reaction will cause a shift to the left. decreasing the rate of the forward reaction will cause a shift to the left. decreasing the rate of the reverse reaction will cause a shift to the right.
Answers: 1

You know the right answer?
Study the electron dot diagrams for lithium, carbon, fluorine, and neon in figure 6-1. choose the st...
Questions

Mathematics, 18.08.2019 13:00


Mathematics, 18.08.2019 13:00

Mathematics, 18.08.2019 13:00

Mathematics, 18.08.2019 13:00

Mathematics, 18.08.2019 13:00


English, 18.08.2019 13:00


Social Studies, 18.08.2019 13:00

Mathematics, 18.08.2019 13:00

Social Studies, 18.08.2019 13:00

Mathematics, 18.08.2019 13:00


Arts, 18.08.2019 13:00

Mathematics, 18.08.2019 13:00


Mathematics, 18.08.2019 13:00


Mathematics, 18.08.2019 13:00



