
Chemistry, 10.12.2019 17:31 erinxmeow8
Balanced complete ionic and net ionic equations
hclo4(aq)+ca(oh)2(aq)→
this is what i got but its wrong h+(aq)+so32−(aq)→so2(g)+h2o(l)
h2so4(aq)+li2so3(aq)→
h+(aq)+so42−(aq)+2li+(aq)+so32−(aq) →2li+(aq)+so42−(aq)+so2(g)+h2o(l)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 01:30
Agas is contained in a thick walled balloon when the pressure changes from 1.21 atm to 2.52 the volume changes from 3.75 l to 1.72 l and the temperature change from 293k to blank k
Answers: 3

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2
You know the right answer?
Balanced complete ionic and net ionic equations
hclo4(aq)+ca(oh)2(aq)→
this is what i g...
hclo4(aq)+ca(oh)2(aq)→
this is what i g...
Questions

English, 20.09.2019 20:00


History, 20.09.2019 20:00


History, 20.09.2019 20:00

Mathematics, 20.09.2019 20:00



Mathematics, 20.09.2019 20:00






Mathematics, 20.09.2019 20:00





English, 20.09.2019 20:00

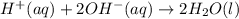
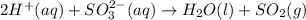
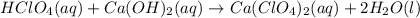
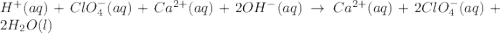
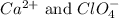 are the spectator ions.
are the spectator ions.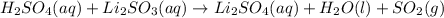
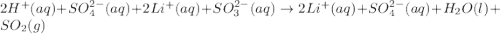
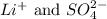 are the spectator ions.
are the spectator ions.

