The acetylene tank contains 35.0 mol c2h2, and the oxygen tank contains 84.0 mol o2.
how many...

Chemistry, 30.08.2019 22:30 Lovely3719
The acetylene tank contains 35.0 mol c2h2, and the oxygen tank contains 84.0 mol o2.
how many moles of co2 are produced when 35.0 mol c2h2 react completely? how do you get 35.0 mol out of c2h2?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Construct the hypothetical phase diagram for metals a and b between room temperature (20c) and 700c, given the following information: * the melting temperature of metal a is 480c. • the maximum solubility of b in a is 4 wt% b, which occurs at 420c. • the solubility of b in a at room temperature is 0 wt% b. • one eutectic occurs at 420c and 18 wt% b–82 wt% a. • a second eutectic occurs at 475c and 42 wt% b–58 wt% a. • the intermetallic compound ab exists at a composition of 30 wt% b–70 wt% a, and melts congruently at 525c.• the melting temperature of metal b is 600c. • the maximum solubility of a in b is 13 wt% a, which occurs at 475c. • the solubility of a in b at room temperature is 3 wt% a.
Answers: 1

Chemistry, 22.06.2019 06:20
If i can still dissolve more sugar into the solution at a certain temperature what would i call that solution
Answers: 3


You know the right answer?
Questions


Mathematics, 23.05.2020 11:58


English, 23.05.2020 11:58

Social Studies, 23.05.2020 11:58

Biology, 23.05.2020 11:58



Mathematics, 23.05.2020 11:58

Mathematics, 23.05.2020 11:58

Social Studies, 23.05.2020 11:58

Advanced Placement (AP), 23.05.2020 11:58



Social Studies, 23.05.2020 11:58

Mathematics, 23.05.2020 11:58



History, 23.05.2020 11:58

Mathematics, 23.05.2020 11:58

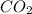 produced are, 70 moles
produced are, 70 moles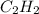 = 35 mole
= 35 mole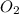 = 84 mole
= 84 mole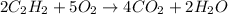
![\farc{4}{2]\times 35=70](/tpl/images/0213/2096/3d3b6.png) moles of carbon dioxide
moles of carbon dioxide

