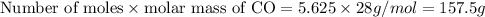Chemistry, 18.09.2019 15:10 emilylittle2442
How many grams of co are needed to react with an excess of fe2o3 to produce (210.3 g fe) 209.7 g fe? show your work.
co (g) + fe2o3 (s) --> fe (s) + co2 (g)

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3

You know the right answer?
How many grams of co are needed to react with an excess of fe2o3 to produce (210.3 g fe) 209.7 g fe?...
Questions

Mathematics, 12.08.2020 07:01


Mathematics, 12.08.2020 07:01

Mathematics, 12.08.2020 07:01

Mathematics, 12.08.2020 07:01




Computers and Technology, 12.08.2020 07:01





Computers and Technology, 12.08.2020 07:01

Mathematics, 12.08.2020 07:01

Mathematics, 12.08.2020 07:01


English, 12.08.2020 07:01

Mathematics, 12.08.2020 07:01

Mathematics, 12.08.2020 07:01

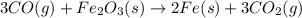
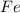 in 210.3 g=
in 210.3 g=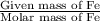
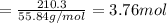
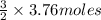 of CO that is 5.64 moles.
of CO that is 5.64 moles.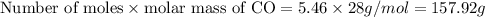
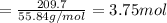
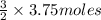 of CO that is 5.625 moles.
of CO that is 5.625 moles.