
Chemistry, 19.04.2021 06:00 AdoreeLayla
S8 +O2 = SO8
Given 80 mols of product, find the amount of each reactant in mols.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 15:30
Plz me ! 1 which of earths spheres contains most of its mass? a atmosphere b hydrosphere c geosphere* d biosphere 2 erosion and weathering are examples of which types of forces? a constructive forces b destructive forces* c gravitational forces d inertia-related forces 3 which of the following statements about earths atmosphere is true? a earths atmosphere contains 78% water vapor which is essentail to life b earths atmosphere contains 21% oxygen c earths atmosphere contains carbon dioxide which all life forms require d earths atmosphere allows radiation from the sun to pass through it and warm earths surface* 4 the strenght of the force of gravity between two objects is determined by which of the following factors? select all that apply a the messes of the objects* b the distance between the objects* c the volumes of the objects d the surface area of the objects 5 earth and moon are kept in there respective orbits due to the influence of a inertia b gravity c gravity and inertia* d neither gravity or inertia if you answer all questions right i will give
Answers: 1

Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1

You know the right answer?
S8 +O2 = SO8
Given 80 mols of product, find the amount of each reactant in mols....
Given 80 mols of product, find the amount of each reactant in mols....
Questions


Mathematics, 09.12.2021 06:00


Physics, 09.12.2021 06:00

Social Studies, 09.12.2021 06:00

History, 09.12.2021 06:00

Mathematics, 09.12.2021 06:00

Mathematics, 09.12.2021 06:00

Computers and Technology, 09.12.2021 06:00




Mathematics, 09.12.2021 06:00

Chemistry, 09.12.2021 06:00

Mathematics, 09.12.2021 06:00

Mathematics, 09.12.2021 06:00


English, 09.12.2021 06:00


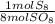 = 10 mol S₈80 mol SO₈ *
= 10 mol S₈80 mol SO₈ * 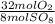 = 320 mol O₂
= 320 mol O₂

