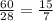Silicon can be made by heating a mixture of sand (silicon dioxide) with magnesium
powder.
The...

Silicon can be made by heating a mixture of sand (silicon dioxide) with magnesium
powder.
The equation for this reaction is shown below.
SiO2 (s)+ 2Mg (s) → 2MgO (s) + Si (s)
Calculate the mass of silicon dioxide needed to make 1 g of silicon.
Relative atomic masses: O = 16; Si = 28
Mass = ?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 21.06.2019 22:30
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1

Chemistry, 22.06.2019 22:30
What must be in balance for temperatures to remain constant?
Answers: 1
You know the right answer?
Questions




History, 03.04.2020 04:05



Mathematics, 03.04.2020 04:05

Computers and Technology, 03.04.2020 04:05





History, 03.04.2020 04:05

History, 03.04.2020 04:05


Mathematics, 03.04.2020 04:05



Medicine, 03.04.2020 04:05