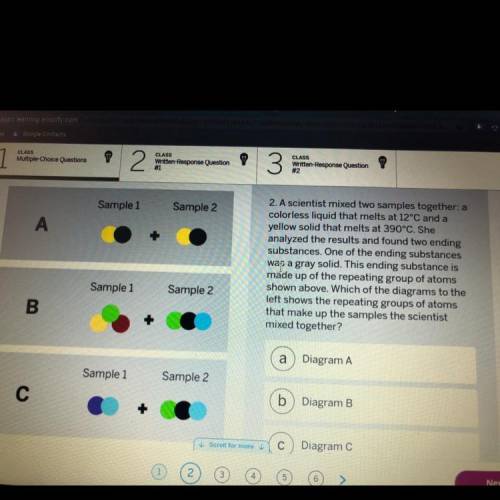
A scientist mixed two samples together: a
colorless liquid that melts at 12°C and a
yellow so...

Chemistry, 15.04.2021 18:10 playaajosh
A scientist mixed two samples together: a
colorless liquid that melts at 12°C and a
yellow solid that melts at 390°С. She
analyzed the results and found two ending
substances. One of the ending substances
was a gray solid. This ending substance is
made up of the repeating group of atoms
shown above. Which of the diagrams to the
left shows the repeating groups of atoms
that make up the samples the scientist
mixed together?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 21.06.2019 18:00
The compound methyl butanoate smells like apples. its percent composition is 58.8% c, 9.9% h, and 31.4% o. what’s the empirical formula ?
Answers: 1


Chemistry, 22.06.2019 19:50
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3
You know the right answer?
Questions


Mathematics, 23.11.2021 01:20

Mathematics, 23.11.2021 01:20

History, 23.11.2021 01:20


Mathematics, 23.11.2021 01:20





Mathematics, 23.11.2021 01:20




Mathematics, 23.11.2021 01:20


Spanish, 23.11.2021 01:20


Mathematics, 23.11.2021 01:20



