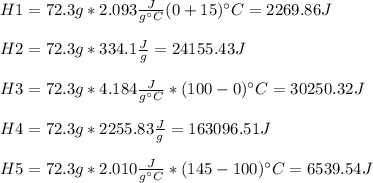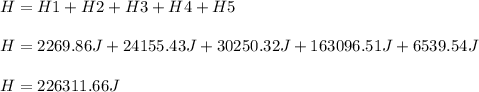Chemistry, 14.04.2021 21:40 deaishaajennings123
72.3 g of ice at -15.0 o C has heat energy added to it until it becomes steam at 145 o C. Calculate the total amount of heat energy needed (in Joules) to accomplish this.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
In which direction will the following reaction go if the standard reduction potentials are 0.80 v for ag/ag+ and –0.44 v for fe/fe2+? ag+ + fe → ag + fe2+ a.)forward b.)the reaction cannot occur. c.) not enough information is given. d.) reverse
Answers: 1

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3
You know the right answer?
72.3 g of ice at -15.0 o C has heat energy added to it until it becomes steam at 145 o C. Calculate...
Questions

English, 16.07.2019 05:40


Biology, 16.07.2019 05:40

Mathematics, 16.07.2019 05:40

Social Studies, 16.07.2019 05:40



Geography, 16.07.2019 05:40

Health, 16.07.2019 05:40

Biology, 16.07.2019 05:40

Social Studies, 16.07.2019 05:40


Physics, 16.07.2019 05:40


English, 16.07.2019 05:40

English, 16.07.2019 05:40

Mathematics, 16.07.2019 05:40


Computers and Technology, 16.07.2019 05:40