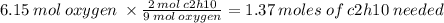Given the following balanced chemical equation:
2C2H10 + 902 +4CO2 + 10H2O
How many moles of...

Chemistry, 14.04.2021 14:00 genyjoannerubiera
Given the following balanced chemical equation:
2C2H10 + 902 +4CO2 + 10H2O
How many moles of C2H10 are needed to completely react with 6.15
moles of oxygen?
O 1.37 mol
2 mol
0277 mol

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:30
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1


Chemistry, 22.06.2019 23:00
What element has similar physical and chemical properties as boron.
Answers: 1
You know the right answer?
Questions



Mathematics, 02.03.2021 16:50

Social Studies, 02.03.2021 16:50

History, 02.03.2021 16:50

Mathematics, 02.03.2021 16:50

Mathematics, 02.03.2021 16:50

Mathematics, 02.03.2021 16:50


Biology, 02.03.2021 16:50

Mathematics, 02.03.2021 16:50

English, 02.03.2021 16:50

Mathematics, 02.03.2021 16:50