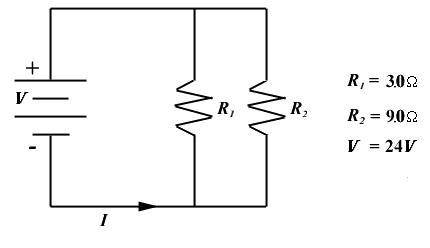
Calculate the power developed in R1.
P1 =_watts
48
190
240
...

Chemistry, 14.04.2021 01:00 zandariouslee
Calculate the power developed in R1.
P1 =_watts
48
190
240
1300

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
Questions


Mathematics, 10.07.2019 21:30


Mathematics, 10.07.2019 21:30



Business, 10.07.2019 21:30



Mathematics, 10.07.2019 21:30



History, 10.07.2019 21:30



Social Studies, 10.07.2019 21:30


English, 10.07.2019 21:30




