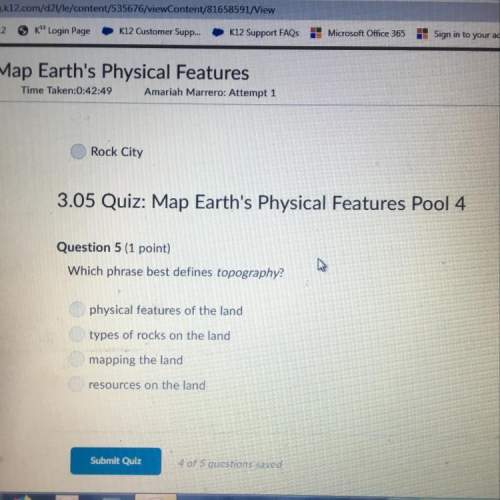
Chemistry, 12.04.2021 21:40 stephanieanaya7
During a titration in the laboratory, 200. mL of 0.600 M HCl (aq) was neutralized with 500. mL RbOH(aq). Determine the molarity of the base.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 01:30
There are main groups in the modern periodic table of elements
Answers: 1

Chemistry, 22.06.2019 18:00
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Chemistry, 23.06.2019 01:30
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3
You know the right answer?
During a titration in the laboratory, 200. mL of 0.600 M HCl (aq) was neutralized with 500. mL RbOH(...
Questions

Mathematics, 23.05.2020 00:02


Mathematics, 23.05.2020 00:02



Mathematics, 23.05.2020 00:02



Biology, 23.05.2020 00:02



Mathematics, 23.05.2020 00:02




Mathematics, 23.05.2020 00:02

History, 23.05.2020 00:02

Social Studies, 23.05.2020 00:02

Law, 23.05.2020 00:02

Social Studies, 23.05.2020 00:02