
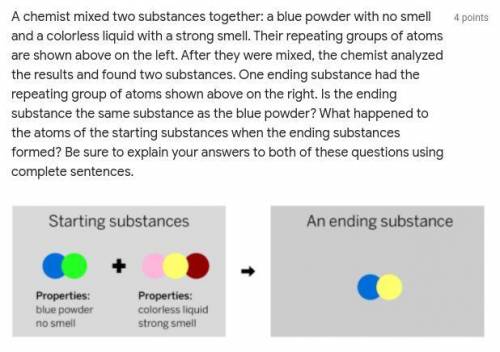
A chemist mixed two substances together: a blue powder with no smell and a colorless liquid with a strong smell. Their repeating groups of atoms are shown above on the left. After they were mixed, the chemist analyzed the results and found two substances. One ending substance had the repeating group of atoms shown above on the right. Is the ending substance the same substance as the blue powder? What happened to the atoms of the starting substances when the ending substances formed? Be sure to explain your answers to both of these questions using complete sentences.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
the mississippians were considered to be horticulturalists, which means they were
Answers: 1

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
A chemist mixed two substances together: a blue powder with no smell and a colorless liquid with a s...
Questions

Chemistry, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01

French, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01


Mathematics, 19.08.2020 01:01

English, 19.08.2020 01:01


Mathematics, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01

Engineering, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01


Mathematics, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01

Mathematics, 19.08.2020 01:01

Law, 19.08.2020 01:01



