Chemistry, 07.04.2021 23:30 brooklynunderwood46
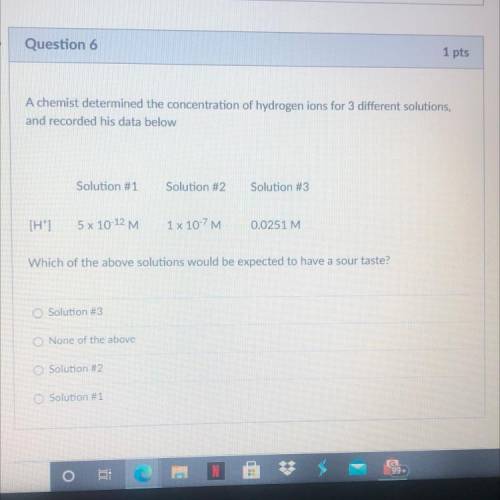
Help pls!! Which of the above solutions would be expected to have a sour taste?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 1

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1
You know the right answer?
Help pls!! Which of the above solutions would be expected to have a sour taste?
...
...
Questions

Health, 30.03.2021 17:00

Mathematics, 30.03.2021 17:00


English, 30.03.2021 17:00





Mathematics, 30.03.2021 17:00


Mathematics, 30.03.2021 17:00

English, 30.03.2021 17:00







Mathematics, 30.03.2021 17:00

Mathematics, 30.03.2021 17:00