
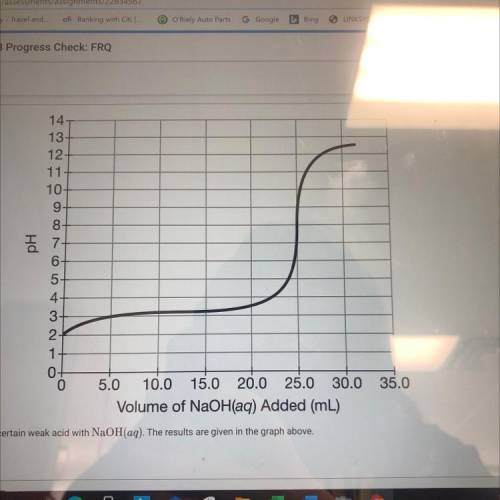
(C) a student ti trates 50.0mL of a 0.1 M solution of a certain weak acid with NaOH(aq). The results are given in the graph above (ii) what specific value from the graph is needed, in addition to the information in part (c) above, to calculate the molar concentration of the NaOH(aq)?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3


Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

You know the right answer?
(C) a student ti trates 50.0mL of a 0.1 M solution of a certain weak acid with NaOH(aq). The results...
Questions


English, 04.02.2021 09:40


History, 04.02.2021 09:40


History, 04.02.2021 09:40


Mathematics, 04.02.2021 09:40

Social Studies, 04.02.2021 09:40

Mathematics, 04.02.2021 09:40

Chemistry, 04.02.2021 09:40



Social Studies, 04.02.2021 09:40

Social Studies, 04.02.2021 09:40


Mathematics, 04.02.2021 09:40


English, 04.02.2021 09:40

Biology, 04.02.2021 09:40



