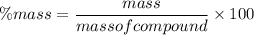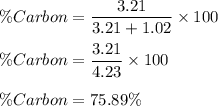Chemistry, 07.04.2021 06:30 xxtonixwilsonxx
A compound is found to be made up of 3.21 g Carbon and 1.02 g Oxygen. Determine the percent composition from this data?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 00:40
Which is a difference between molecular compounds and ionic compounds? select the correct answer below: question 5 options: molecular compounds typically form between a metal and a nonmetal, while ionic compounds typically form between nonmetals. molecular compounds result from the transfer of electrons between atoms to form ions, while ionic compounds result from the sharing of electrons between neutral atoms. molecular compounds are formed of discrete, neutral molecules, while ionic compounds are formed of large repeating arrays of opposite charges. molecular compounds have high melting points and high boiling points, while ionic
Answers: 3

Chemistry, 22.06.2019 07:00
This image is an example of a(n) a) atom. b) compound. c) mixture. d) molecule.
Answers: 1

Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2
You know the right answer?
A compound is found to be made up of 3.21 g Carbon and 1.02 g Oxygen. Determine the percent composit...
Questions





Mathematics, 04.12.2020 01:20


Mathematics, 04.12.2020 01:20

Mathematics, 04.12.2020 01:20


Mathematics, 04.12.2020 01:20


Mathematics, 04.12.2020 01:20



Mathematics, 04.12.2020 01:20

Mathematics, 04.12.2020 01:20



Mathematics, 04.12.2020 01:20

Chemistry, 04.12.2020 01:20