

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:50
Calculate the number of molecules present in 0.750 mol of mgo.
Answers: 3

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 23.06.2019 00:30
What are the advantages of using the metric system? designed as a decimal system making conversions simpler more accurate system of measurement has prefixes that correspond to an amount to use with all base units used by the entire scientific community
Answers: 2

You know the right answer?
A buffer is made with 0.493 M of the weak base methylamine () with 0.493 M (KA = 5.6 x 10^-4)
What...
Questions

Mathematics, 14.10.2021 16:20

History, 14.10.2021 16:20


Advanced Placement (AP), 14.10.2021 16:30



Social Studies, 14.10.2021 16:30


Social Studies, 14.10.2021 16:40

English, 14.10.2021 16:40


History, 14.10.2021 16:40



Social Studies, 14.10.2021 16:50

Biology, 14.10.2021 16:50


Mathematics, 14.10.2021 16:50

English, 14.10.2021 16:50

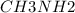 ) with 0.493 M
) with 0.493 M 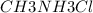 (KA = 5.6 x 10^-4)
What is the pH of this buffer system?
(KA = 5.6 x 10^-4)
What is the pH of this buffer system?

