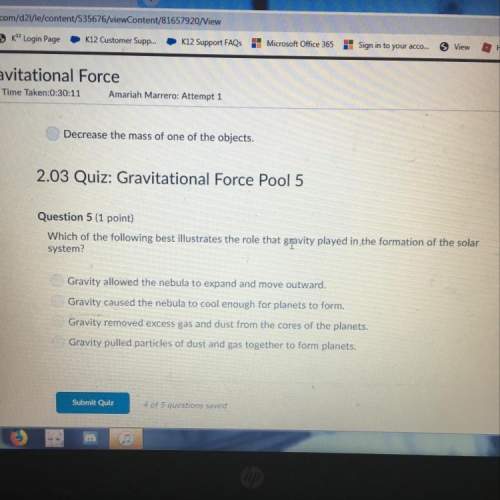
Based on Table H, what is the vapor pressure of CH3COOH and 90. °C?
1.
40. kPa
2.
...

Chemistry, 06.04.2021 09:00 Kimbeeeerly4569
Based on Table H, what is the vapor pressure of CH3COOH and 90. °C?
1.
40. kPa
2.
48 kPa
3.
114 kPa
4.
150. kPa

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1


Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1

Chemistry, 23.06.2019 00:30
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1
You know the right answer?
Questions

Business, 30.11.2020 06:40


Mathematics, 30.11.2020 06:40


Mathematics, 30.11.2020 06:40

History, 30.11.2020 06:50






Mathematics, 30.11.2020 06:50



Biology, 30.11.2020 06:50


History, 30.11.2020 06:50

Social Studies, 30.11.2020 06:50

Mathematics, 30.11.2020 06:50