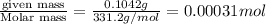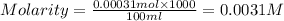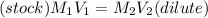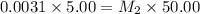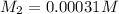Chemistry, 02.04.2021 03:00 kseniyayakimov
A stock solution was created by adding 0.1042 g of lead (II) nitrate to a 100.00 mL volumetric flask and diluting to volume with deionized water. A diluted solution was then created by removing 5.00 mL of the stock solution and placing into into a 50.00 mL volumetric flask and then diluting to volume with deionized water. What is the concentration (in molarity, M) of the diluted solution

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
About 70 percent of the earth's surface is water-covered, and about 96.5 percent of all earth's water is salt water. identify the watery feature on earth that is made of freshwater rather than salt water. a) bay b) glacier c) ocean d) sea it is not incomplete this is the true question
Answers: 1

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2
You know the right answer?
A stock solution was created by adding 0.1042 g of lead (II) nitrate to a 100.00 mL volumetric flask...
Questions

Mathematics, 05.12.2020 04:30

History, 05.12.2020 04:30

Physics, 05.12.2020 04:30




Social Studies, 05.12.2020 04:30


History, 05.12.2020 04:30

History, 05.12.2020 04:30

Mathematics, 05.12.2020 04:30

Mathematics, 05.12.2020 04:40

Mathematics, 05.12.2020 04:40

Mathematics, 05.12.2020 04:40




Mathematics, 05.12.2020 04:40


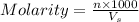
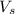 = volume of solution in ml
= volume of solution in ml
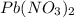 =
=