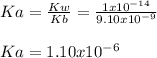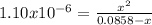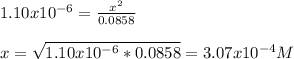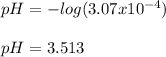Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Strong conductivity of plasma allows it to act and react as and
Answers: 2

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
You know the right answer?
Calculate the pH at the equivalence point when 22.0 mL of 0.200 M hydroxylamine, HONH2, is titrated...
Questions



History, 13.07.2019 18:20

Mathematics, 13.07.2019 18:20

Biology, 13.07.2019 18:20

Biology, 13.07.2019 18:20

History, 13.07.2019 18:20

History, 13.07.2019 18:20


History, 13.07.2019 18:20

History, 13.07.2019 18:20


Biology, 13.07.2019 18:20


Biology, 13.07.2019 18:20

Social Studies, 13.07.2019 18:20

Biology, 13.07.2019 18:20

Social Studies, 13.07.2019 18:20

Biology, 13.07.2019 18:20

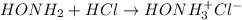
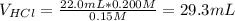
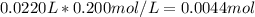
![[HONH_3^+Cl^-]=\frac{0.0044mol}{0.022L+0.0293L} =0.0858M](/tpl/images/1235/6200/36779.png)

![Ka=\frac{[H_3O^+][HONH_2]}{[HONH_3^+]}](/tpl/images/1235/6200/cfc73.png)