
Chemistry, 01.04.2021 19:20 bryanmcmillianjr
A student titrates an unknown weak acid, HA, to a pale pink phenolphthalein endpoint with 25.0 mL of 0.100 M NaOH. The student then titrates in 11.0 mL of 0.100 M HCl to the solution containing the neutralized unknown acid (HCl was delivered from a second buret). The pH of the resulting solution is pH

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3



Chemistry, 23.06.2019 02:30
Which words or phrases identify layers of groundwater? check all that apply. water table kettle lake saturation zone underground lake sinkhole will give brainiest, answer quickly.
Answers: 1
You know the right answer?
A student titrates an unknown weak acid, HA, to a pale pink phenolphthalein endpoint with 25.0 mL of...
Questions

Mathematics, 24.06.2019 13:00

Mathematics, 24.06.2019 13:00


History, 24.06.2019 13:00

Mathematics, 24.06.2019 13:00



Mathematics, 24.06.2019 13:00



English, 24.06.2019 13:10



Physics, 24.06.2019 13:10



Social Studies, 24.06.2019 13:10

Mathematics, 24.06.2019 13:10

Mathematics, 24.06.2019 13:10

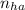 = 2.5 mmoles ------ 1
= 2.5 mmoles ------ 1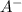 + HCL ---------> HA + C
+ HCL ---------> HA + C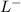
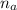 = 1.1 mmoles --------- 2
= 1.1 mmoles --------- 2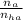 = 1.1 / 2.5 ≈ 1/2 ( this show that when the pH = 4.7 approximately half of the A^- have been converted to HA )
= 1.1 / 2.5 ≈ 1/2 ( this show that when the pH = 4.7 approximately half of the A^- have been converted to HA ) 

