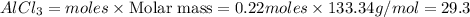PLEASE HELP
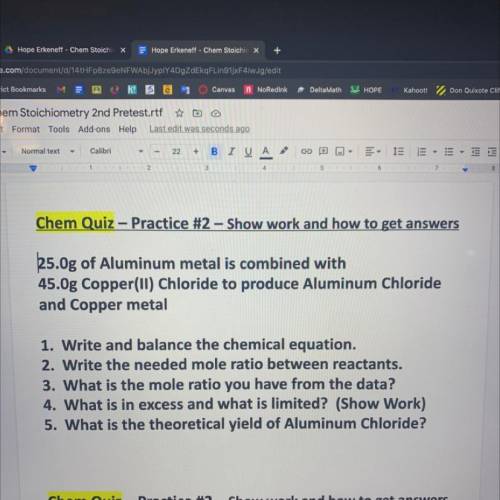
25.0g of Aluminum metal is combined with
45.Og Copper(ll) Chloride to produce Aluminum Chloride
and Copper metal
1. Write and balance the chemical equation.
2. Write the needed mole ratio between reactants.
3. What is the mole ratio you have from the data?
4. What is in excess and what is limited? (Show Work)
5. What is the theoretical yield of Aluminum Chloride?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:10
Between 2014 and 2016, more than 25,000 children in flint, michigan, drank water that was contaminated with lead from lead pipes. during this time, the city claimed the water was safe to drink. which of these actions could the city have taken to ensure that the drinking water was free from lead?
Answers: 3

Chemistry, 22.06.2019 10:10
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3

Chemistry, 22.06.2019 14:10
Precision can be defined as the o exact center of a data set. o reproducibility of a measured value. o correlation between two variables that are measured in a data set agreement between a measured value and an accepted value.
Answers: 2

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2
You know the right answer?
PLEASE HELP
25.0g of Aluminum metal is combined with
45.Og Copper(ll) Chloride to produce Alu...
45.Og Copper(ll) Chloride to produce Alu...
Questions



Mathematics, 23.05.2021 02:50

English, 23.05.2021 02:50

Mathematics, 23.05.2021 02:50

Mathematics, 23.05.2021 02:50

Mathematics, 23.05.2021 02:50

Mathematics, 23.05.2021 02:50

History, 23.05.2021 02:50





Mathematics, 23.05.2021 02:50

Mathematics, 23.05.2021 02:50

Mathematics, 23.05.2021 02:50

Chemistry, 23.05.2021 02:50



Mathematics, 23.05.2021 02:50

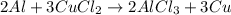
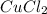 : 2 moles of
: 2 moles of 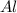
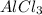 is 29.3 g
is 29.3 g 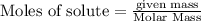
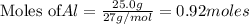
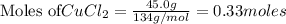
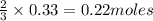 of
of