
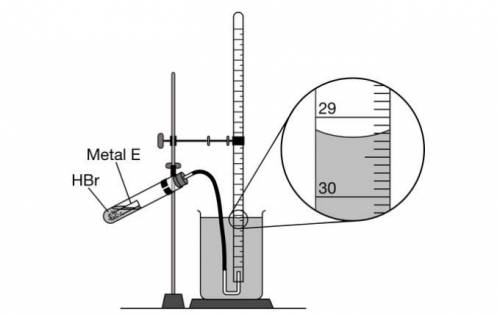
As shown in the following diagram, 10 mL samples of HBr(aq) are added to different quantities of E(s) in a closed test tube. At the start of each trial, the gas-collecting tube is filled completely with distilled water. The samples of E(s) react with HBr(aq) , and the H2(g) produced is collected in the gas-collecting tube.
The enlarged view of the gas-collecting tube at the end of trial 3 is shown in the diagram.
What should the student record as the volume, in mL , of H2(g) collected in trial 3?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1
You know the right answer?
As shown in the following diagram, 10 mL samples of HBr(aq) are added to different quantities of E(s...
Questions

English, 15.12.2021 17:30


English, 15.12.2021 17:30

Mathematics, 15.12.2021 17:30

Mathematics, 15.12.2021 17:30



Physics, 15.12.2021 17:30

Mathematics, 15.12.2021 17:30






Chemistry, 15.12.2021 17:30



English, 15.12.2021 17:30

Mathematics, 15.12.2021 17:30

Mathematics, 15.12.2021 17:30



