
Chemistry, 30.03.2021 01:00 kamkam1713
A gas tank contains 2.5 moles of oxygen, 7.0 moles of nitrogen and 0.5 mole of water vapor. The total pressure of the mixture is 852 torr. Calculate the partial pressure of each gas. Show work.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2


Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

You know the right answer?
A gas tank contains 2.5 moles of oxygen, 7.0 moles of nitrogen and 0.5 mole of water vapor. The tota...
Questions


History, 10.04.2020 03:34

Chemistry, 10.04.2020 03:35


Biology, 10.04.2020 03:35


History, 10.04.2020 03:35

Mathematics, 10.04.2020 03:35









Mathematics, 10.04.2020 03:35


Mathematics, 10.04.2020 03:35

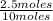 = 0.25mole fraction of nitrogen =
= 0.25mole fraction of nitrogen = 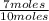 = 0.7mole fraction of water vapor =
= 0.7mole fraction of water vapor = 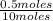 = 0.05
= 0.05

