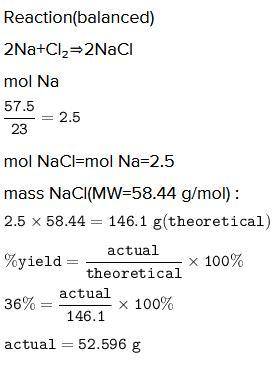Chemistry, 29.03.2021 19:20 joyceslater16
A chemist reacted 57.50 grams of sodium metal with an excess amount of chlorine gas. The chemical reaction that occurred is shown.
Na + Cl2 → NaCl
If the percentage yield of the reaction is 86%, what is the actual yield? Show your work, including the use of stoichiometric calculations and conversion factors.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Each pair of clay balls represents to planetesimals if each plane test molluscum pound of the same material and is separated by the same distance which pair experiences the greatest gravitational attraction
Answers: 2

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2


Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
A chemist reacted 57.50 grams of sodium metal with an excess amount of chlorine gas. The chemical re...
Questions


History, 26.08.2019 13:30




History, 26.08.2019 13:30


Geography, 26.08.2019 13:30


Social Studies, 26.08.2019 13:30



Physics, 26.08.2019 13:30



English, 26.08.2019 13:30

Mathematics, 26.08.2019 13:30



Social Studies, 26.08.2019 13:30