
Chemistry, 29.03.2021 16:50 lindamillscotton90
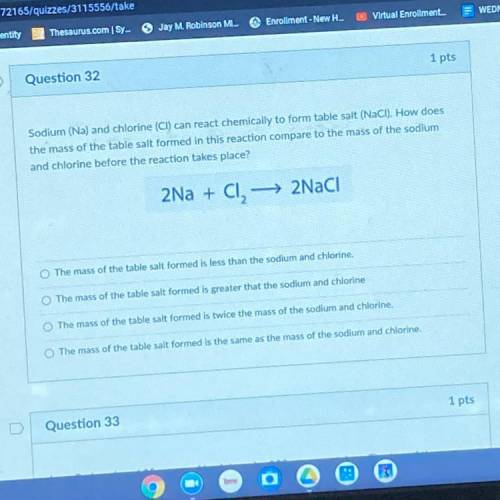
Sodium (Na) and chlorine (CI) can react chemically to form table salt (NaCl). How does the mass of the table salt formed in this reaction compare to the mass of the sodium and chlorine before the reaction takes place?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Write a paragraph at least 10 sentences explaining how rocks are used today in society
Answers: 1

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
You know the right answer?
Sodium (Na) and chlorine (CI) can react chemically to form table salt (NaCl). How does
the mass of...
Questions

Spanish, 17.11.2020 22:00

Mathematics, 17.11.2020 22:00

Mathematics, 17.11.2020 22:00

Mathematics, 17.11.2020 22:00

Biology, 17.11.2020 22:00

Mathematics, 17.11.2020 22:00



Mathematics, 17.11.2020 22:00


Mathematics, 17.11.2020 22:00



Mathematics, 17.11.2020 22:00

Mathematics, 17.11.2020 22:00


Physics, 17.11.2020 22:00


Mathematics, 17.11.2020 22:00



