
Chemistry, 28.03.2021 22:20 pamdhuber4224
NEED HELP ASAP
A concept map for four types of intermolecular forces and a certain type of bond is shown.
Compare the relative strength of the two forces B and C. Explain how you determined this comparison by identifying the forces.
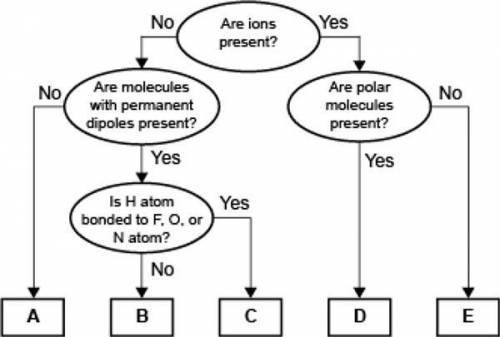

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1
You know the right answer?
NEED HELP ASAP
A concept map for four types of intermolecular forces and a certain type of bond is...
Questions


History, 12.07.2019 09:30

Mathematics, 12.07.2019 09:30


Social Studies, 12.07.2019 09:30

Mathematics, 12.07.2019 09:30

History, 12.07.2019 09:30

English, 12.07.2019 09:30

Social Studies, 12.07.2019 09:30

Mathematics, 12.07.2019 09:30

Social Studies, 12.07.2019 09:30

Health, 12.07.2019 09:30



Mathematics, 12.07.2019 09:30

Biology, 12.07.2019 09:30

Business, 12.07.2019 09:30


Business, 12.07.2019 09:30



