2C2H6 + 702 + 4CO2 + 6H2O
How many grams of water will be produced if you start
with 92.92 gr...

Chemistry, 26.03.2021 19:40 littleprinces
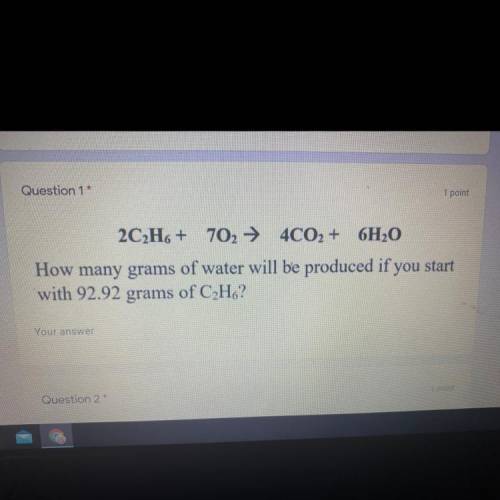
2C2H6 + 702 + 4CO2 + 6H2O
How many grams of water will be produced if you start
with 92.92 grams of C2H6?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
Questions


Geography, 13.11.2020 21:00

Mathematics, 13.11.2020 21:00


History, 13.11.2020 21:00

Social Studies, 13.11.2020 21:00

Mathematics, 13.11.2020 21:00

English, 13.11.2020 21:00


Health, 13.11.2020 21:00



Mathematics, 13.11.2020 21:00


Social Studies, 13.11.2020 21:00

Mathematics, 13.11.2020 21:00

History, 13.11.2020 21:00

Physics, 13.11.2020 21:00




