
Chemistry, 26.03.2021 17:00 amirasaleh1012
Can someone help me solve the last two lines of this table. Please include explanation. Thanks
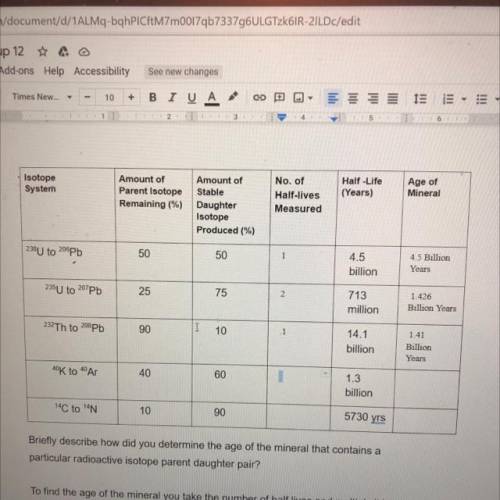

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2
You know the right answer?
Can someone help me solve the last two lines of this table. Please include explanation. Thanks
Questions

Mathematics, 03.08.2020 14:01






Mathematics, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01




English, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01



History, 03.08.2020 14:01


Mathematics, 03.08.2020 14:01




