Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3


Chemistry, 22.06.2019 17:20
Which of these features are formed when hot groundwater is forced out through cracks in the earth's surface?
Answers: 2
You know the right answer?
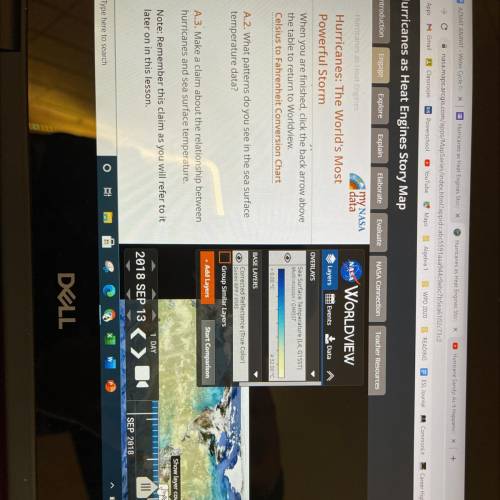
What patterns do you see in the sea surface temperature data? Hurricanes worlds most powerful storm...
Questions


English, 14.10.2019 08:01


Chemistry, 14.10.2019 08:01

Social Studies, 14.10.2019 08:01

Mathematics, 14.10.2019 08:01

Mathematics, 14.10.2019 08:01


History, 14.10.2019 08:01

Mathematics, 14.10.2019 08:01

Mathematics, 14.10.2019 08:01