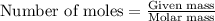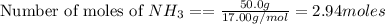Calculate the precise Molar Mass of NH3 and calculate the moles in 50.0
grams....

Chemistry, 26.03.2021 08:10 kevinhernandez582
Calculate the precise Molar Mass of NH3 and calculate the moles in 50.0
grams.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 23.06.2019 11:50
Charles's law describes the relationship of the volume and temperature of gas at a constant mass and pressure. according to this law, what would happen to the temperature of the gas if its volume decreased from 1.0 l to 0.50 l?
Answers: 3
You know the right answer?
Questions




Advanced Placement (AP), 23.04.2021 20:20

History, 23.04.2021 20:20

Social Studies, 23.04.2021 20:20

History, 23.04.2021 20:20


Mathematics, 23.04.2021 20:20



Mathematics, 23.04.2021 20:20

Mathematics, 23.04.2021 20:20


Mathematics, 23.04.2021 20:20

Mathematics, 23.04.2021 20:20




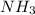 is 17.00 g and moles in 50.0 grams is 2.94.
is 17.00 g and moles in 50.0 grams is 2.94.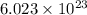 of particles and weigh equal to its molecular mass.
of particles and weigh equal to its molecular mass.