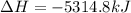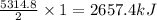Chemistry, 26.03.2021 01:00 turboslayer
Butane C4H10 (g),(Hf = –125.7), combusts in the presence of oxygen to form CO2 (g) (Delta. Hf = –393.5 kJ/mol), and H2O(g) (Delta. Hf = –241.82) in the reaction: 2 upper C subscript 4 upper H subscript 10 (g) plus 13 upper O subscript 2 (g) right arrow 8 upper C upper O subscript 2 plus 10 upper H subscript 2 upper O (g). What is the enthalpy of combustion, per mole, of butane? Use Delta H r x n equals the sum of delta H f of all the products minus the sum of delta H f of all the reactants.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Mrs. smith ordered a root beer float (vanilla ice cream + root beer). mrs. smith noticed that the three states of matter (solid, liquid, and gas) all existed simultaneously in her root beer float. a. identify each phase of matter in the root beer float. b. describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? ) c. identify one phase change you would see in a root beer float and described what causes this change.
Answers: 2

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1


Chemistry, 23.06.2019 02:30
Apound is approximately 0.45 kilogram. a persons weighs 87 kilograms. what is the persons’s weight, in pounds, when expressed to the correct number of significant figures
Answers: 1
You know the right answer?
Butane C4H10 (g),(Hf = –125.7), combusts in the presence of oxygen to form CO2 (g) (Delta. Hf = –393...
Questions

Mathematics, 11.10.2019 06:30

Mathematics, 11.10.2019 06:30


Mathematics, 11.10.2019 06:30


Mathematics, 11.10.2019 06:30

Chemistry, 11.10.2019 06:30

Computers and Technology, 11.10.2019 06:30


Mathematics, 11.10.2019 06:30



Mathematics, 11.10.2019 06:30


Mathematics, 11.10.2019 06:30

Business, 11.10.2019 06:30


Mathematics, 11.10.2019 06:30

History, 11.10.2019 06:30

English, 11.10.2019 06:30

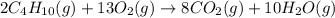
![\Delta H=[n\times H_f_{products}]-[n\times H_f_{reactants}]](/tpl/images/1221/8923/4f68b.png)
![\Delta H=[8\times H_f_{CO_2}+10\times H_f_{H_2O}]-[2\times H_f_{C_4H_{10}+13\times H_f_{O_2}}]](/tpl/images/1221/8923/e94db.png)
![\Delta H=[(8\times -393.5)+(10\times -241.82)]-[(2\times -125.7)+(13\times 0)]](/tpl/images/1221/8923/8343f.png)