
Chemistry, 26.03.2021 01:00 kaliyab191
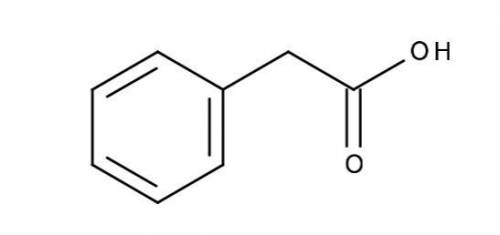
Calculate (a) the pH of a 0.0250 M solution of phenylacetic acid, and (b) the pH of a 0.0500 M solution of sodium phenylacetate. The pKa of phenylacetic acid is 4.31, and its structure is shown below.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:20
Which statement accurately describes the relationship between air pressure, air density, or altitude? as altitude increases, pressure increases.as altitude increases, air density increases.air pressure and density are lowest at sea level.denser air exerts more pressure than less dense air.
Answers: 2

Chemistry, 22.06.2019 21:20
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1

Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3

Chemistry, 23.06.2019 03:00
What happens in the particles of a gas when the gas is compressed
Answers: 1
You know the right answer?
Calculate (a) the pH of a 0.0250 M solution of phenylacetic acid, and (b) the pH of a 0.0500 M solut...
Questions

History, 27.01.2021 01:00

Mathematics, 27.01.2021 01:00

Mathematics, 27.01.2021 01:00

Advanced Placement (AP), 27.01.2021 01:00

Mathematics, 27.01.2021 01:00

Arts, 27.01.2021 01:00




English, 27.01.2021 01:00

Mathematics, 27.01.2021 01:00


Mathematics, 27.01.2021 01:00



Mathematics, 27.01.2021 01:00


Mathematics, 27.01.2021 01:00




