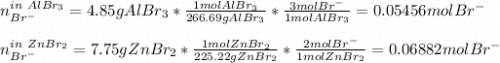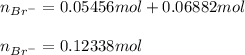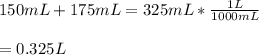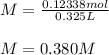Chemistry, 25.03.2021 21:50 ambercuevas2707
Two different bromide solutions are mixed with each other: Solution 1 is an aqueous solution of 4.85 g aluminum bromidein 150. ml water and solution 2 has a volume of 175 ml and contains 7.75 g of zinc bromide. You mix the two solutions together in a large beaker. What is the bromide concentration in moles/L in the mixture

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
What mass of carbon dioxide is produced from the complete combustion of 4.50×10−3 g of methane?
Answers: 2

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1
You know the right answer?
Two different bromide solutions are mixed with each other: Solution 1 is an aqueous solution of 4.85...
Questions

Spanish, 04.05.2021 01:00



Mathematics, 04.05.2021 01:00

SAT, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00

Chemistry, 04.05.2021 01:00



English, 04.05.2021 01:00




English, 04.05.2021 01:00

Social Studies, 04.05.2021 01:00

English, 04.05.2021 01:00

Arts, 04.05.2021 01:00

Physics, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00