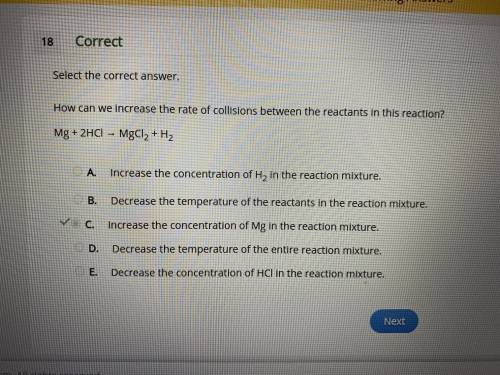
How can we increase the rate of collisions between the reactants in this reaction?
Mg + 2HCl → MgCl2 + H2
A.
Increase the concentration of H2 in the reaction mixture.
B.
Decrease the temperature of the reactants in the reaction mixture.
C.
Increase the concentration of Mg in the reaction mixture.
D.
Decrease the temperature of the entire reaction mixture.
E.
Decrease the concentration of HCl in the reaction mixture.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 21.06.2019 22:30
Explain why scientists use shared characteristics to make cladograms.
Answers: 1

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1

You know the right answer?
How can we increase the rate of collisions between the reactants in this reaction?
Mg + 2HCl → MgCl...
Questions


Mathematics, 18.10.2021 23:20

Mathematics, 18.10.2021 23:20


SAT, 18.10.2021 23:20

Business, 18.10.2021 23:20




Mathematics, 18.10.2021 23:30





Mathematics, 18.10.2021 23:30