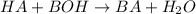Chemistry, 25.03.2021 05:50 YourFinalsAreHere
Suppose that you are performing a titration on a monoprotic acid. Titration of this monoprotic acid required a volume of 0.1L of the base being used. If the same volume of a diprotic acid would instead have been used (same volume of a diprotic acid versus the originally used monoprotic acid), and if we assume furthermore that the concentration of the diprotic acid and the monoprotic acids are equal, how does the volume of base required to reach the endpoint for the diprotic acid compare to the volume of base required to reach the endpoint for the monoprotic acid? A) More base is likely required to reach the endpoint for the diprotic acid than for the monoprotic acid under these conditionsB) Less base is likely required to reach the endpoint for the diprotic acid than for the monoprotic acid. C) Exactly the same amount of base is required for both monoprotic and diprotic acids under the situations described here. D) A completely different indicator will need to be used here.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1
You know the right answer?
Suppose that you are performing a titration on a monoprotic acid. Titration of this monoprotic acid...
Questions



History, 03.12.2021 18:30


Mathematics, 03.12.2021 18:30


Physics, 03.12.2021 18:30

Mathematics, 03.12.2021 18:30


Arts, 03.12.2021 18:30

Biology, 03.12.2021 18:30


SAT, 03.12.2021 18:30

Mathematics, 03.12.2021 18:30


Computers and Technology, 03.12.2021 18:30




 has a valency of 2.
has a valency of 2.