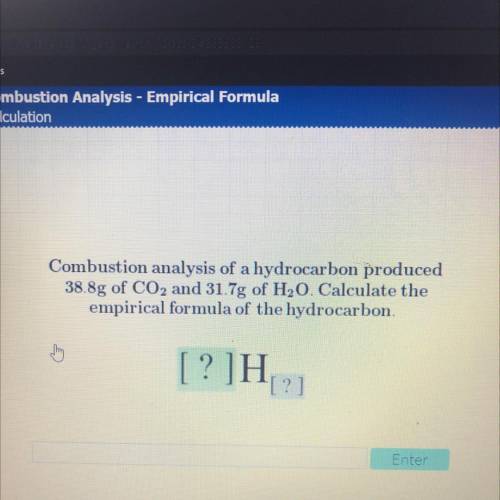
Combustion analysis of a hydrocarbon produced
38.8g of CO2 and 31.7g of H20. Calculate the
em...

Chemistry, 24.03.2021 18:30 caudhdi11721
Combustion analysis of a hydrocarbon produced
38.8g of CO2 and 31.7g of H20. Calculate the
empirical formula of the hydrocarbon.
[?]H
[?]

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 08:30
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

You know the right answer?
Questions

Mathematics, 10.10.2019 23:20

Mathematics, 10.10.2019 23:20

English, 10.10.2019 23:20





Mathematics, 10.10.2019 23:20



Mathematics, 10.10.2019 23:20

Mathematics, 10.10.2019 23:20




Mathematics, 10.10.2019 23:20


Chemistry, 10.10.2019 23:30


Business, 10.10.2019 23:30



