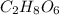Chemistry, 24.03.2021 04:30 taniyahreggienae
A compound has an empirical formula of c3h4o3. It's molar mass is 180 g. What is the molecular formula?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3


Chemistry, 23.06.2019 05:50
Which of the following is not a characteristic of s waves?
Answers: 1
You know the right answer?
A compound has an empirical formula of c3h4o3. It's molar mass is 180 g. What is the molecular formu...
Questions

Mathematics, 03.04.2020 15:29








Computers and Technology, 03.04.2020 15:30





Mathematics, 03.04.2020 15:31


Chemistry, 03.04.2020 15:31